
From the medical industry and beyond, durable, anti-corrosive aluminum is essential to performance. Aluminum is used extensively for components in various applications because of its unique combination of properties – including that it is especially well-suited to anodizing.
Here, we’ll discuss the features and benefits of aluminum, the advantages and limitations of anodization, and how AL-COAT® biocompatible chromium coatings provide a versatile alternative to achieving anti-corrosive properties for a wide variety of applications.
Aluminum: Versatile, All-Around Team Player of Metals
Like an athlete with a comprehensive skill set and superior performance in their respective sport, aluminum receives high marks for its adaptability and versatility. The metal is prized for teamwork — aluminum’s unique properties play well across diverse applications — for many reasons:
Strong corrosion resistance
Aluminum naturally forms a thin, protective oxide layer that prevents the metal from interacting with environmental elements. This property grants it resistance against corrosion, primarily when it’s alloyed with other elements or coated.
Lightweight
Due to its low density, aluminum components are lighter than those made from other metals. Aluminum is an excellent choice in automotive and aerospace applications, where weight reduction can lead to fuel savings and improved performance.
Malleable and Ductile
Aluminum is easy to shape, form, and machine — you can roll it into thin sheets and draw it into wires in various products.
Electrical and Thermal Conductivity
Aluminum is an excellent conductor of both electricity and heat, making it ideal for wire, connectors, and heat exchangers.
Reflective
Aluminum can reflect both light and radiant heat.
Compatible
Aluminum can be alloyed with other elements, including copper, magnesium, silicon, and zinc, to enhance its mechanical properties. Also, unlike other metals that become brittle, aluminum retains its toughness at low temperatures.
Cost-effective
Never underestimate one of aluminum’s most essential qualities: providing the best balance of strength, weight, and cost.
Recyclable
Aluminum is 100% recyclable without any loss of its natural properties; in fact, around 80% of aluminum production in the United States is from recycled content. This eco-friendly aspect supports a circular economy and reduces the need for mining new raw materials.
Aluminum and Anodization
Anodizing is an electrochemical process that converts the metal surface into a durable, corrosion-resistant, anodic oxide finish. Aluminum is especially well-suited to anodization due to some of the properties we previously mentioned, and the benefits conferred in the actual anodizing process — protective, natural oxide formation, a porous oxide layer, and good thermal conductivity.
The anodizing process involves immersing the aluminum in an acid electrolyte bath and passing an electric current through it – this invokes oxidation, producing aluminum oxide. Here’s the anodizing process, simplified in three steps:
- Clean: First, remove any contaminants and debris from the aluminum instrument.
- Anodize: Connect the clean aluminum instrument to the positive side of an electrical circuit (the anode). The electrolyte, often sulfuric acid, is the medium for transferring ions and electric current. Once the power is on, the electrolyte releases oxygen ions, which combine with the aluminum atoms on the instrument’s surface, forming a layer of aluminum oxide.
- Seal: Seal microscopic pores on the anodized aluminum instrument via boiling water to increase corrosion resistance.
Advantages of Anodizing Process
We already mentioned some of the benefits of anodization, but let’s take a closer look.
Enhanced durability
Anodized surfaces are hard and highly resistant to abrasion and wear. As a result, anodized aluminum is suitable for various applications, including aerospace, automotive, construction, and consumer products.
Improved aesthetics
Based on the application’s requirements, anodized aluminum is available in different finishes to increase or decrease reflective properties. Furthermore, it’s possible to dye anodized aluminum to achieve different colors, and you can also apply decorative patterns or logos to the surface.
Corrosion- and heat-resistant
The anodic oxide structure originates from the aluminum substrate and is composed entirely of aluminum oxide, ensuring a tight bond. The oxide layer is a barrier between the underlying aluminum and the environment. Finally, anodized aluminum has a high melting point and can withstand harsh chemicals and poor weather conditions better than untreated aluminum.
Reduced maintenance
Compared to untreated aluminum, anodized aluminum requires minimal maintenance. It retains its appearance and protective properties for extended periods, reducing the need for frequent cleaning or refinishing.
Non-toxic
Anodizing is an environmentally friendly, non-toxic process that doesn’t involve heavy metals or hazardous chemicals. In addition to medical applications, you’ll find anodized aluminum in everything from aerospace components to kitchen utensils.
Anodizing Limitations
Working through the design process with qualified engineers and metallurgists is critical, as some limitations to anodization exist.
Color limitations
While you can choose a matte or shiny finish and the color of anodized aluminum, achieving precise and consistent color can be challenging. Also, specific colors can fade when exposed to UV light and sunlight.
Thickness variability
Besides variations in color matching, there may also be some inconsistency in anodized layer thickness across different instruments. Anodizing bath chemistry, temperature, and current density all affect layer thickness.
Complex shapes
Instruments with challenging shapes, as well as those with internal cavities, can make anodization more difficult. The electrolyte bath and subsequent sealing need to access all component areas; this is harder to achieve with intricate parts.
Not suitable for all aluminum alloys
Some alloys may not produce a consistent or attractive finish when anodized.
Wear over time
While anodized surfaces are hard and resistant to wear, they can still be scratched or damaged by abrasive materials or sharp objects. Once the anodized layer is breached, the underlying aluminum can be susceptible to corrosion.
Alternative to Anodizing: Introducing AL-COAT®
A licensed product and process developed and managed by the Armoloy Corporation laboratories, AL-COAT® is a biocompatible, pure metallic chromium explicitly engineered for aluminum. When it comes to aluminum components, AL-COAT® offers distinct advantages over traditional anodizing:
Corrosion defense
Previous testing from an independent laboratory showed that AL-COAT® coating increases the corrosion resistance of 6061-T6 aluminum and remains adherent when exposed to many environments, especially to alkaline and chlorinated solutions. AL-COAT’s corrosion resistance makes it well-suited for components exposed to such environments, reducing the need for frequent maintenance and replacement. The table below illustrates corrosion resistance of AL-COAT® over hard coat anodizing in harsh environments for 6061-T6 aluminum.
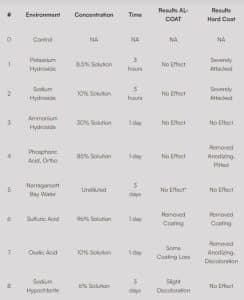
Table 1. Improved corrosion resistance of the Al-Coat over hard Coat Anodizing in harsh environments.
Metallurgical, Materials Science & Mechanical Engineering, Failure Analysis, Testing & Design.*
*Al-Coat accelerated galvanic corrosion in the bare aluminum
Increased Wear Resistance
The higher hardness of AL-COAT® provides better wear resistance, making it ideal for components subjected to friction, contact, or abrasive conditions, thereby extending their expected service life.
Uniform Surface Finish
AL-COAT’s uniform chrome coating creates a smooth surface, reducing the risk of surface imperfections that can impact performance in precision machined components.
Lower Friction
The low coefficient of friction of AL-COAT® facilitates smoother sliding or movement of machined parts, reducing energy losses and enhancing overall efficiency.
Both anodizing and AL-COAT® are effective surface treatment methods for aluminum substrates, each offering unique characteristics and applications. While anodizing is versatile and suitable for various industries, AL-COAT® excels in providing superior corrosion resistance, hardness, and low friction. For machined components made from aluminum, AL-COAT® emerges as a potentially superior choice, ensuring extended durability and optimal performance in demanding environments.